
Myocarditis rates at 25X in teens is apparently ‘reassuring’!
If you want to join the debate on our articles, comments are open and welcome on our Substack version, so please feel free to subscribe and join the conversation.
Last month HART editors wrote a slightly humorous article entitled How to get published in the Covid era. Humorous, that is, if it wasn’t so serious.
One of the main recommendations for would-be authors was to make your abstract conclusion fit the ‘Vaccines Safe & Effective’ narrative, regardless of your findings. We gave a number of examples, including a prospective study on myocarditis from Thailand which showed 1 in 43 adolescents with clinical or subclinical myo- or pericarditis and concluded: ‘adolescents receiving mRNA vaccines should be monitored for cardiovascular side effects.’
This week a national study from New Zealand topped the lot in terms of the discordance between their findings and their conclusion. The team looked at national data for 12 predetermined adverse events of special interest (AESIs) between 1-21 days following the Pfizer BioNTech covid vaccine, and compared them with background rates from 2014-2019. They found that for 5-19-year-olds, the risk of cardiac inflammation was increased 25-fold. This is likely to have been an underestimate as they excluded any events that occurred on the same date as vaccination (day 0) to avoid counting events that occurred prior to vaccination (another tip in our article was omitting data that might worsen your results). It has already been noted that a significant number of people develop symptoms on the day of vaccination, as shown here.
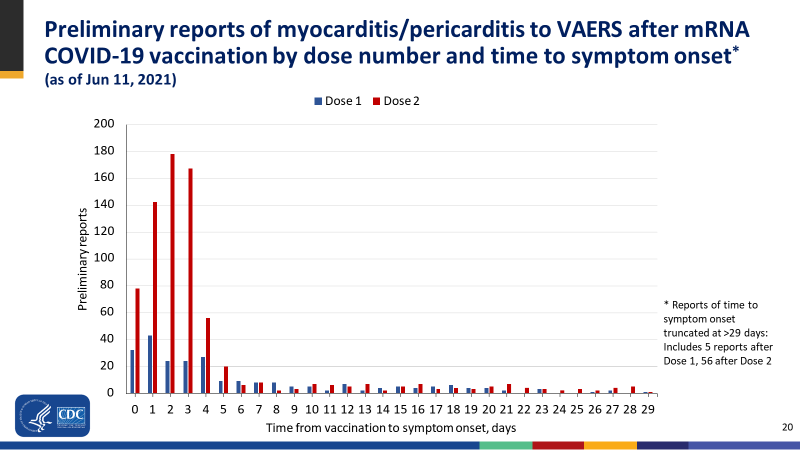
Figure 1. from CDC COVID-19 Vaccine Task Force June 2021
In the New Zealand study, the increased risk of myo-/pericarditis was highest in the 5-19s (standardised incidence ratio (SIR) 25.6 [15.5–37.5]) but was also raised in 20-39s (SIR 6.6 [5.1–8.2]) and 40-59s (3.4 [2.5–4.4]). The only other adverse event of special interest signal was for cutaneous vasculitis in the 20-19 age group (3.7 [1.1– 7.0]).
Of course the problem with myocarditis and the inverse link to age, has been recognised for more than 2 years, as highlighted in the same CDC paper:
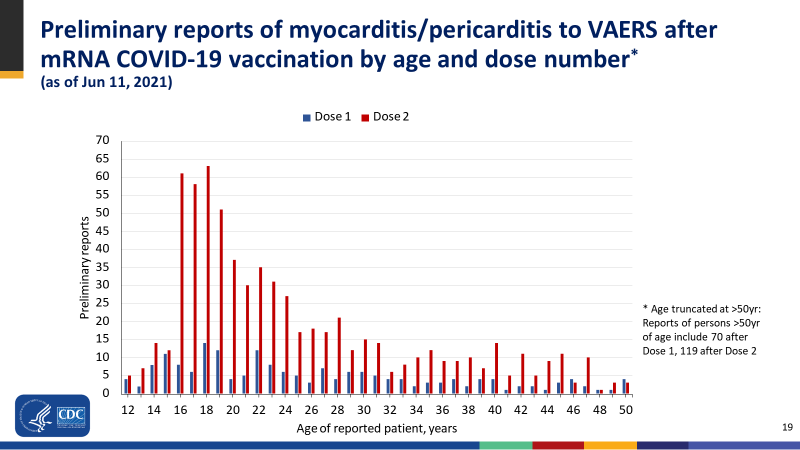
Figure 2. from CDC COVID-19 Vaccine Task Force June 2021
So their conclusion:
‘A statistically significant association between BNT162b2 vaccination and myo/pericarditis was observed. This association has been confirmed internationally. BNT162b2 was not found to be associated with the other AESIs investigated, except for SOCV following the first dose of BNT162b2 in the 20–39 years age group only, providing reassurances around the safety of the vaccine’.
Who exactly is meant to be reassured by these data ⎼ the vaccine injured New Zealanders and their families, the international academics whose previous findings are confirmed or the investors of money and reputation in the vaccines? The reader can decide but the HART team has no doubt it is not the vaccine injured.
